Understanding CDMO Regulatory Support for Global Filings
Global regulatory filings (IND/CTA, NDA/MAA, ANDA) demand extensive CMC documentation and compliance.
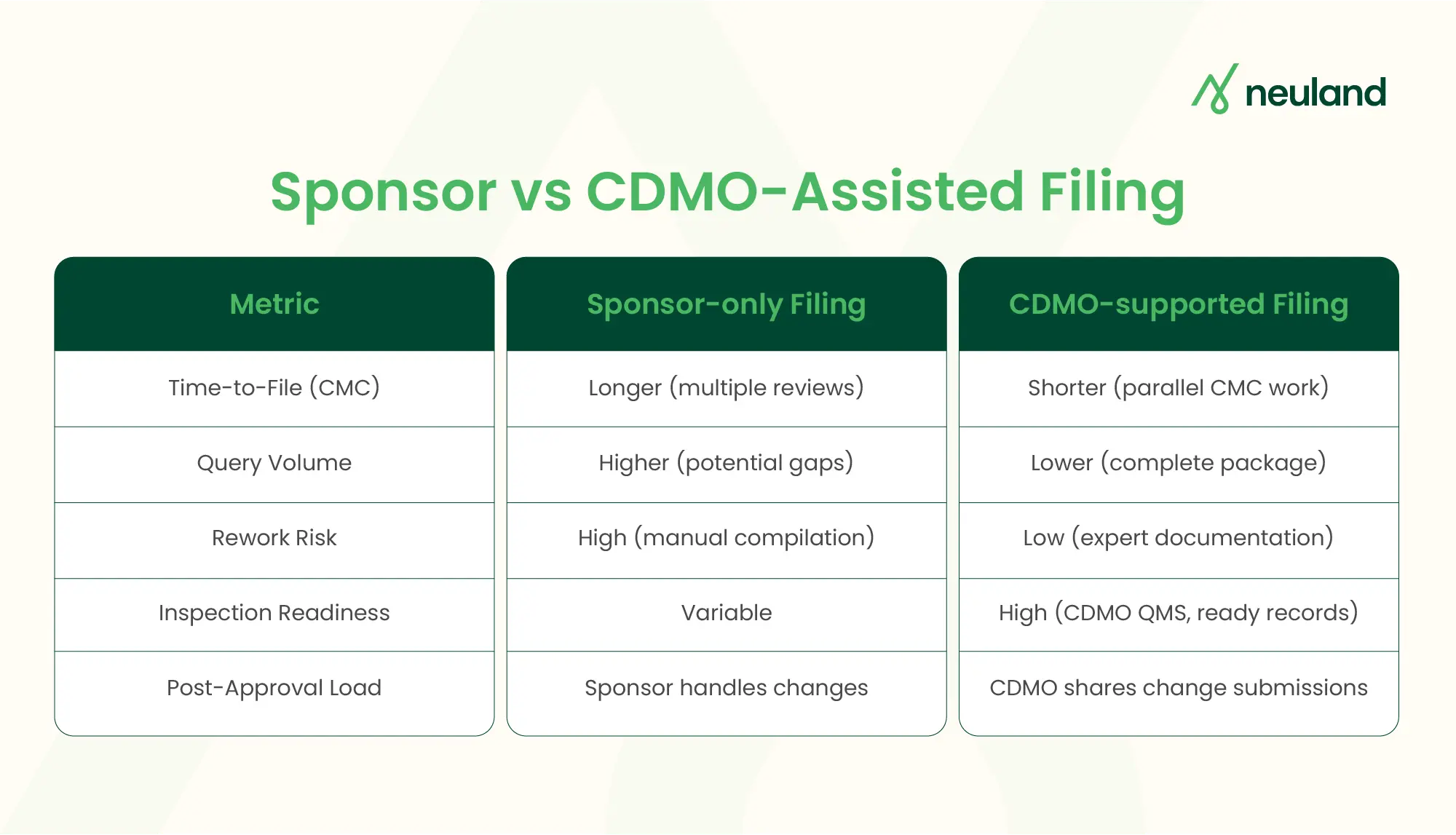
An experienced CDMO like Neuland not only manufactures APIs/peptides but also prepares Module 3 CMC packages, stability data, and regulatory dossiers. With deep expertise, a CDMO can support filings “right-first-time,” reducing sponsor workload and regulatory queries.
Evidence shows strong CDMO engagement accelerates approvals and minimizes rework. In this report, we detail what CDMO regulatory support entails, compare sponsor-only vs CDMO-assisted filings, outline evaluation criteria, and provide a sourcing checklist.

CDMO Regulatory Support: Crucial Role in Filings
A CDMO with regulatory expertise compiles the entire Chemistry section of submissions. This includes:
- Process Documentation: Complete process flow, control strategies, and impurity profiles for Drug Master Files or CTD Module 3.
- Analytical Data: Method validation reports and assay development needed for IND/NDA.
- Stability Data: Ongoing stability study results to justify shelf-life (often incomplete at IND but supported in later filings).
- CMC Narrative: A coherent CMC summary linking development to manufacturing outcomes.
Neuland’s track record highlights this capability: we have enabled 25 INDs and 4 NDAs by furnishing drug substance for each milestone. Partnering CDMOs are accustomed to CTD dossier preparation and writing documents to FDA/EMA standards.
CDMOs ensure high-quality documentation and on-time submissions. With Neuland’s support, sponsors get submissions that align with CMC regulatory requirements across regions.
Regulatory Work Delegated to CDMOs
Specifically, a CDMO can handle:
- IND Submission Requirements: CMC sections, batch certificates, and initial stability specs. Often sponsors reference CDMO Drug Master File (DMF) to avoid repetitive chemistry details.
- NDA/MAA/ANDA Support: Full CMC dossier writing, including full purification/analytical reports and validation protocols. The CDMO provides commercial-scale batch summaries, validation of critical assays, and expanded stability data.
- Global Submissions: Adaptation of CMC modules to region-specific formats (e.g., US FDA vs EMA eCTD, Japanese filings, etc.). CDMOs gather foreign regulatory documents (e.g., CEP/EDQM certificates, WHO PDEs) as needed.
- Regulatory Queries & Post-Approval: Assisting in addressing CMC questions during review (e.g., additional data requests), and later submissions like site additions, process changes, or new stability results.
Checklist: Evaluating CDMO Regulatory Support
Sponsors should vet CDMOs on:
- Filing Experience: Proven record of supporting INDs, NDAs, ANDAs, MAAs. (E.g., Neuland cites dozens of filings.)
- Documentation Expertise: In-house regulatory affairs or CMC authors skilled in Module 3/CTD structure.
- Global Knowledge: Understanding of region-specific guidelines (FDA, EMA, PMDA, etc.) and dossier variations.
- Quality Systems: Robust quality assurance, eCTD publishing systems, and archiving procedures.
- Responsiveness: Defined process for query-response and post-approval submissions.
A tick-list approach (e.g., “Has CDMO led prior filings?”, “Is there a regulatory team interface?”) helps ensure alignment with sponsor needs.
From Submission Support to Approval Confidence
True CDMO regulatory support is measured by how seamlessly a partner translates development work into submission-ready CMC documentation, aligned with evolving CMC regulatory requirements, and defensible under inspection.
It is about anticipating regulator questions before they are asked, aligning technical decisions with the NDA submission process, and maintaining disciplined control through post-approval regulatory support.
Regulatory success is not driven by volume of documents—it is driven by coherence, consistency, and quality culture. For Regulatory Affairs and CMC leaders, the right regulatory support CDMO does more than compile data. It integrates process understanding, validation rigor, and inspection readiness into a unified strategy that reduces deficiency letters, minimizes clarification cycles, and strengthens overall compliance standards.
Neuland’s integrated approach—spanning development, documentation, and lifecycle change management—positions it to support global filings with technical discipline and operational accountability.
If you are preparing an upcoming submission, now is the time to discuss your regulatory and CMC requirements about structuring a filing-ready strategy.
FAQs
|
|
|
|