CDMO API Manufacturing for Complex Molecules
What is API manufacturing in pharma?
API (Active Pharmaceutical Ingredient) manufacturing is the process of producing the biologically active component in drugs through chemical synthesis. It converts raw materials into high-purity medicinal compounds, which are later combined with excipients to create finished drugs.
However, the gap between a synthesis that works at lab scale and one that delivers consistent GMP batches at commercial volume is where most pharma programs run into trouble.
That's why CDMO API manufacturing has become the default path for companies working on complex drug candidates. Peptides, high-potency APIs, multi-chiral compounds, and molecules requiring hazardous chemistries all demand capabilities that most sponsors can't justify building in-house.
This article breaks down what makes a molecule "complex," how CDMOs support API manufacturing, and what to look for when choosing a partner.
What Makes an API "Complex" in Pharmaceutical Manufacturing
An API qualifies as complex when its synthesis requires specialized equipment, tight stereochemical control, or hazardous reagent handling. The industry breaks these molecules into a few broad categories:
- High-potency APIs (HPAPIs) active at microgram doses, requiring containment down to OEB 5 levels
- Peptide APIs that are built through sequential amino acid coupling, where each step introduces potential yield loss
- Multi-chiral-center molecules where enantiomeric purity must be tightly controlled
- Hazardous chemistry products involving cryogenic reactions, high-pressure hydrogenation, bromination, or fluorination
Each category demands infrastructure that goes well beyond standard organic chemistry. Cryogenic reactors need massive cooling systems. Hydrogenation requires high-pressure vessels. HPAPI production needs multi-tier containment. Very few CDMOs maintain all these capabilities at GMP standard under one roof.
This is exactly why complex molecule synthesis has become one of the fastest-growing segments in contract API manufacturing.
Download Neuland’s CDMO Services Brochure |
How CDMOs Support Complex Molecule Synthesis
A capable CDMO pharma partner does more than run reactions. It provides the full stack of services needed to move a complex molecule from candidate selection through commercial supply.
Route Scouting and API Process Development
API process development starts with evaluating multiple synthetic routes. The shortest laboratory route is rarely the best one for commercial production. A good CDMO weighs each option against cost, scalability, impurity control, and safety.
Design of Experiments (DoE) methodology helps identify the optimal parameters for each reaction step. This reduces the risk of surprises at scale and builds the data package regulators expect.
Scale-Up and Technology Transfer
Moving a process from lab to pilot to commercial scale introduces variables that chemistry alone can't predict. Heat transfer efficiency drops in larger reactors. Mixing behavior changes. Exothermic reactions that are easy to manage in a flask can become dangerous in a 10,000-liter vessel.
CDMOs with scale-up experience bring pattern recognition that reduces failed batches. They know which steps are most sensitive to equipment changes and how to adjust parameters without compromising quality.
GMP API Manufacturing Under ICH Q7
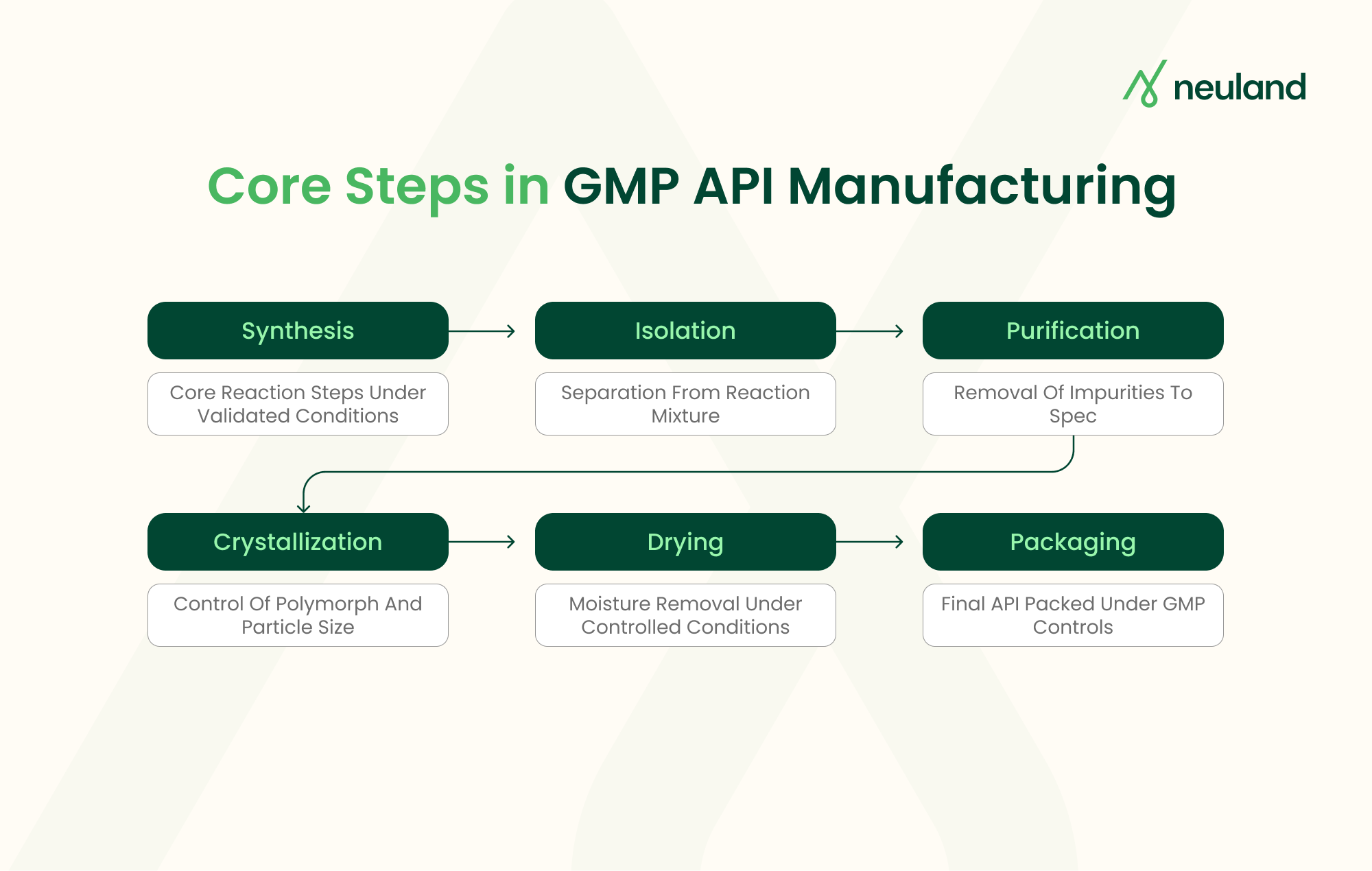
GMP API manufacturing follows a structured workflow. Starting materials are qualified. Reactions are run under validated conditions. Intermediates are tested. The final API is isolated, purified and packaged under controlled environments.
Each stage follows ICH Q7 guidelines, which the FDA treats as the foundation for API compliance. CDMO API manufacturing includes synthesis, isolation, purification, crystallization, drying, and packaging, with in-process controls at every transition.
Crystallization is especially important. It controls polymorphic form, particle size, and downstream processability. A poorly designed crystallization step can compromise bioavailability or stability, even when the chemistry upstream is perfect.
Why Pharma Companies Opt For CDMO API Manufacturing
The shift toward outsourcing isn't a short-term trend. It reflects structural changes in how drug development works.
Building a GMP facility with specialized equipment costs $200 million or more. That capital is hard to justify when a CDMO can provide the same capability without the fixed overhead. Emerging biotech firms typically lack the infrastructure entirely. And even large pharma companies are reducing their in-house complex chemistry footprint.
According to Grand View Research, the global API CDMO market reached $107 billion in 2024 and is projected to nearly double by 2033. CDMO API manufacturing accounts for the majority of pharmaceutical services.
The regulatory environment is also pushing companies toward specialized partners. FDA CDER warning letters jumped 50% in FY2025, with most targeting manufacturing facilities that failed GMP inspections. CDMOs with clean inspection records directly lower that risk for sponsors.
A few other reasons stand out:
- Specialized expertise that takes years to build internally
- Regulatory filing support across DMFs, INDs, and NDAs
- Flexible capacity that scales from clinical to commercial
- Speed to market through ready infrastructure instead of multi-year facility builds
Peptide API manufacturing is a clear example. Demand for GLP-1 drugs like semaglutide created capacity pressure that no single pharma company could solve alone. CDMOs stepped in with dedicated peptide platforms, and several have committed hundreds of millions to expansion.
What to Look for in a CDMO for Complex API Manufacturing

Only a subset of contract partners are equipped to execute complex molecule synthesis effectively. When evaluating CDMOs, a few factors separate strong candidates from average ones.
Technical Range
Can the CDMO handle your chemistry today and your next program too? A partner skilled in small molecule API manufacturing may not have peptide capabilities. Look for breadth across multiple modalities, including HPAPI containment, cryogenic chemistry, and hydrogenation.
Regulatory Track Record
Ask how many DMFs and regulatory filings the CDMO has supported. A clean FDA inspection history is the most reliable quality signal. Partners with multi-agency approvals (FDA, EMA, PMDA) reduce friction when filing in multiple regions.
Development-to-Commercial Continuity
Partners that offer process development, clinical API manufacturing, and commercial supply at the same site reduce technology transfer risk. Mid-program switches add months of rework and regulatory documentation.
Quality System Maturity
Strong quality systems cover deviation handling, change control, validated computerized records, and robust CAPA programs. These are non-negotiable for complex molecule programs, where minor process changes can trigger new impurities.
Choosing the Right Partner for Complex API Programs
Complex API programs need a collaborator with deep technical experience, a clean regulatory record, and the infrastructure to scale from lab to commercial production without compromise.
Neuland Laboratories brings this kind of depth to CDMO API manufacturing. With three cGMP-certified facilities, over 360 R&D scientists, and capabilities spanning complex small molecules, peptides, and high-potency APIs, Neuland supports pharma and biotech clients across the full development lifecycle.
Their regulatory approvals from the FDA, EMA, and PMDA, combined with a growing peptide platform built for commercial-scale production, make them a strong fit for sponsors with complex chemistry needs.
For teams planning their next complex molecule program, choosing the right partner can shape both the timeline and the outcome. Talk to Neuland's team today.
FAQ's
|
|
|
|
|