What Is API Process Development? Key Steps and Best Practices
Turning a drug candidate into a viable therapy requires developing a process to produce that molecule at scale.
Active pharmaceutical ingredient (API) process development is the discipline of designing and optimizing the manufacturing process for the API. It takes a lab-scale concept and prepares it for efficient, large-scale production.
This article will explore what API process development entails, its key stages, how it differs from API manufacturing, and best practices for success.
What Is API Process Development?
API process development involves creating a production process for an active pharmaceutical ingredient that consistently delivers quality, purity, and yield.
This involves optimizing reactions, selecting raw materials and solvents, defining equipment, and setting critical parameters. Crucially, the process must be efficient and cost-effective, and built under good manufacturing practices and ready for validation.
By the end of this, the team has established a validated API production process that can be executed repeatedly to produce the API with consistent quality attributes.
This foundation, in turn, supports later stages of drug development by ensuring the active pharmaceutical ingredient manufacturing process is reproducible and compliant.
Key Stages in API Process Development
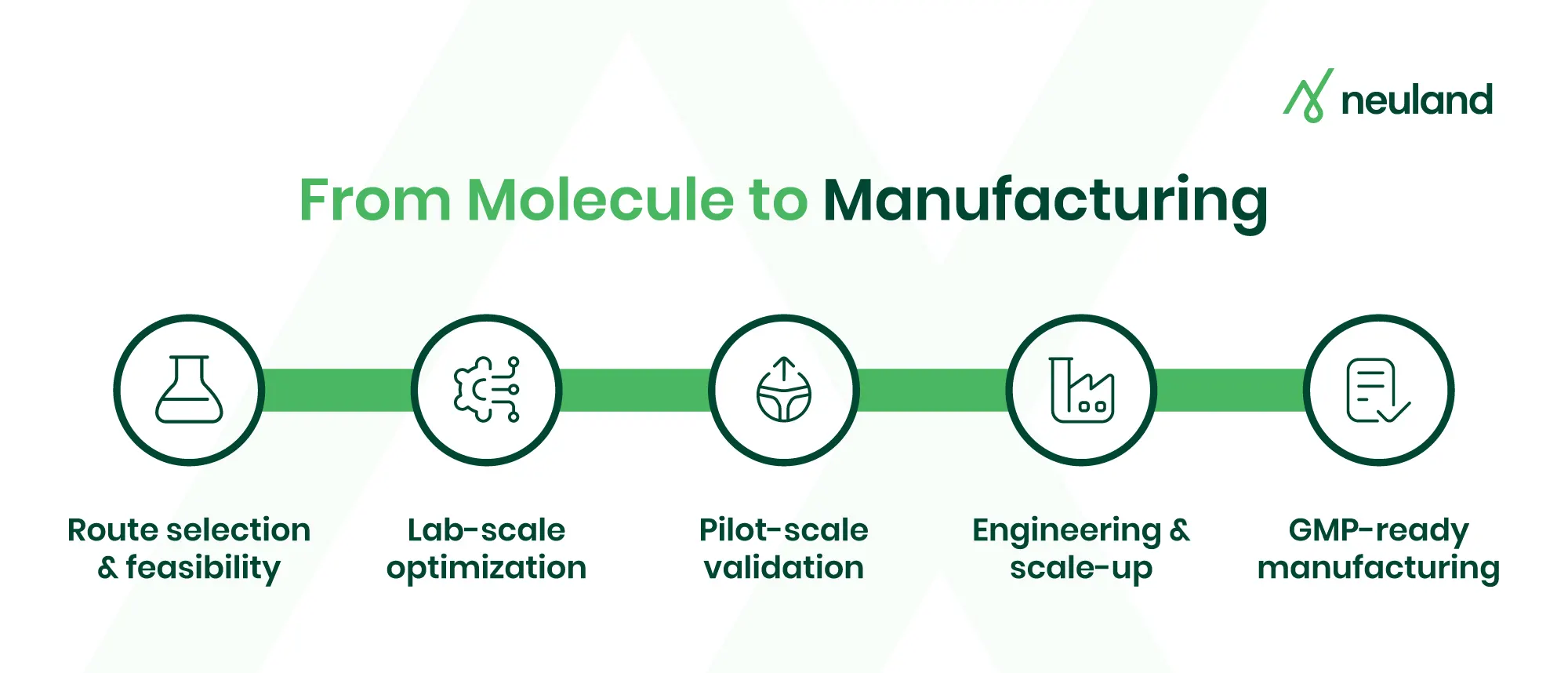
Projects progress through a series of well-defined stages, each building on the last to reduce risk and prepare for scale-up.
At each step, researchers refine the process to improve yield and ensure safety. The major steps in API process development typically include:
- Route Scouting & Lab Feasibility
Chemists test various small-scale synthetic routes to find feasible API production pathways, assessing yields, raw material availability, safety, and scalability. Early experiments identify the most promising route and address safety and efficiency concerns. - Process Optimization
Once a route is chosen, the process is optimized in the lab. Developers tweak conditions to improve yield, purity, and cost. They reduce steps or bottlenecks. By this phase's end, the team has a more efficient, reproducible procedure and understands key process parameters. - Pilot Scale Demonstration
The optimized process is tested at pilot scale to evaluate performance in plant-mimicking equipment. This stage uncovers scale-dependent challenges, like changes in mixing and heat transfer that affect reactions. Pilot runs help fine-tune control strategies and verify that critical process parameters (CPPs) stay within control limits at larger volumes. - Engineering & Scale-Up Batches
With pilot data, engineering batches are performed in production-scale reactors to verify the process in the actual plant environment. This step, often part of tech transfer, checks equipment fit and operational practicality. These batches identify and resolve issues, such as adapting to different agitator designs or cleaning requirements, before full production. - GMP Qualification Batches
Finally, the process is run under GMP conditions to produce qualification or validation batches. These batches are used to demonstrate consistent quality and are often used in regulatory submissions. Process validation studies are done during this stage, producing multiple batches to confirm reliable large-scale performance.
API Process Development vs API Manufacturing
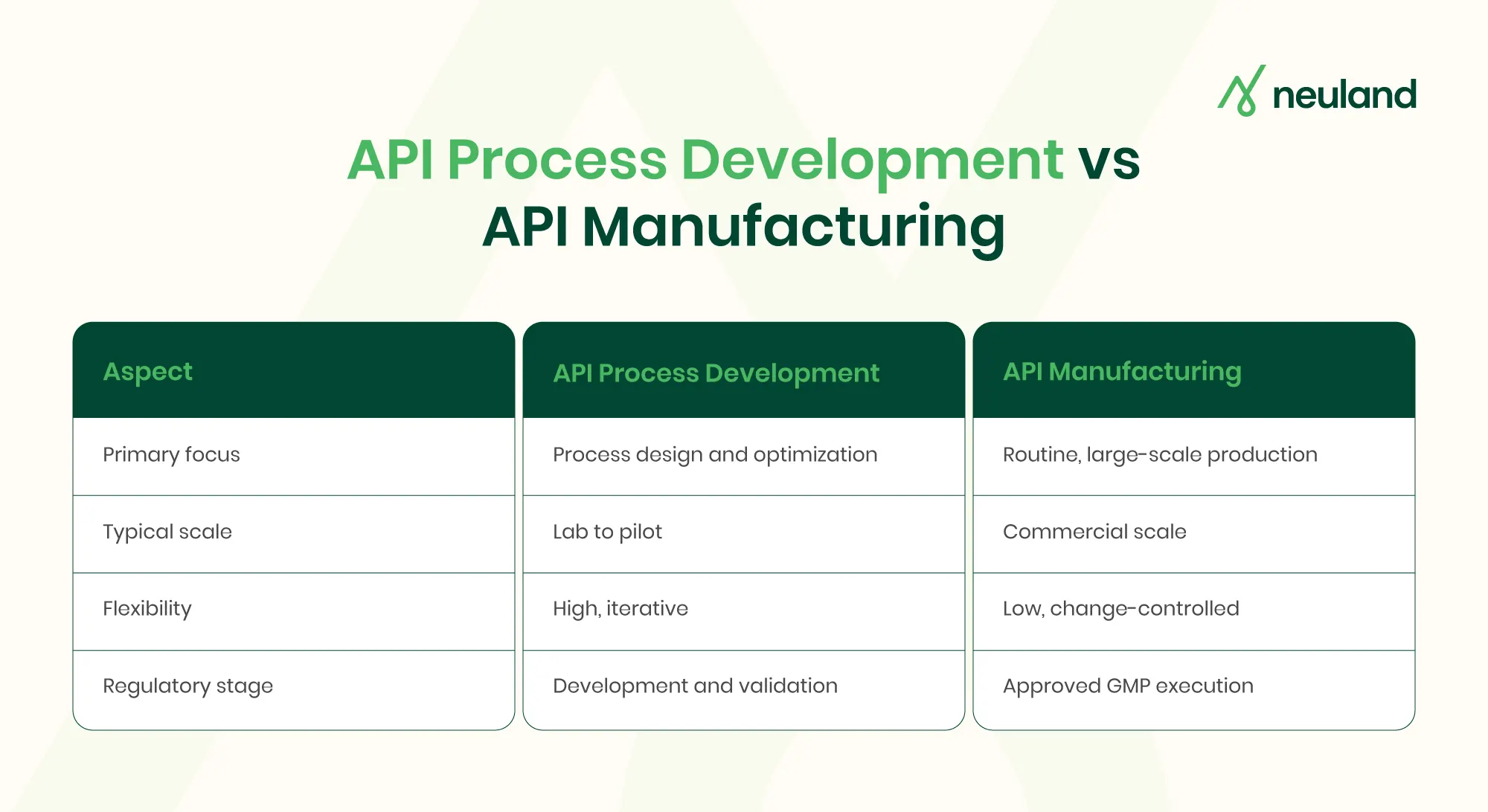
API process development is an R&D-driven phase focused on designing, refining, and proving a reliable process for making the API. It is iterative and exploratory, involving lab experiments and small-scale trials that gradually scale up. The outcome is a validated, regulatory-ready process.
API manufacturing, by contrast, is the execution of that established process at production scale under strict GMP conditions to produce the API in commercial quantities. Development allows optimization and adjustments, while manufacturing permits only tightly controlled changes to ensure consistency.
The two phases are closely linked. Knowledge flows from development to manufacturing through tech transfer, and early GMP batches are often produced while development is still ongoing.
Before launch, the process undergoes formal validation to demonstrate consistent performance at scale. Once validated, API manufacturing enters steady-state production, delivering the active pharmaceutical ingredient reliably and at volume.
Choosing the Right API Process Development Partner
API process development is rarely executed in isolation. As molecules progress from discovery to scale, pharma and biotech firms often depend on external partners for expertise, infrastructure, and support. The effectiveness of this partnership can directly influence timelines, cost control, and regulatory readiness.
The right partner combines scientific depth with practical scale-up experience, ensuring that early process decisions translate smoothly into a reliable API manufacturing process. This is especially critical as development progresses from laboratory optimization to pilot and GMP environments, where poor handoffs or knowledge gaps can introduce costly delays.
At a minimum, an effective API process development partner should demonstrate:
- Proven experience with complex chemistries, including a strong track record of moving APIs from early development through scale-up
- Regulatory maturity, with systems aligned to global expectations such as the US FDA, EMA, and PMDA
- End-to-end process understanding, spanning development, scale-up, and technology transfer
- Strong collaboration and project governance, enabling transparent communication and faster issue resolution
In practice, many organizations engage a CDMO at this stage because the role demands integrated development and manufacturing expertise.
Neuland Labs’ Approach to API Process Development
Neuland Labs exemplifies this partner-led model. With more than four decades of experience, Neuland has supported the development of complex small molecules and peptides, guiding APIs from early-stage process design through commercial manufacturing.
At Neuland, API process development begins in the R&D laboratory, where processes are built using rigorous scientific and engineering methodologies.
Development teams apply Quality by Design (QbD) principles, structured risk management, and Design of Experiments (DoE) to systematically understand process behavior, identify critical parameters, and establish robust control strategies early.
This data-driven approach allows potential scale-up risks to be addressed upfront, rather than discovered during manufacturing. By integrating development, scale-up, and regulatory readiness from the outset, Neuland ensures that processes are not only efficient at lab scale but also reproducible, compliant, and commercially viable.
Check out how Neuland supports API process development, which leads to life-changing medicines reaching patients faster and more reliably.
FAQs
|
|
|
|