Dual Sourcing in CDMO Partnerships: A Strategic Framework for Resilient Drug Supply
Pharma companies face growing supply-chain risks (geopolitical, quality, demand spikes) that can stall clinical trials and delay commercialization.
Dual sourcing CDMOs – engaging two qualified contract manufacturing partners for the same product – emerges as a key risk mitigation strategy to ensure continuity and flexibility.
This blog outlines when and how to implement dual sourcing versus single or multiple sourcing, and describes the governance needed, including contracts, tech transfer, quality agreements, KPIs, and oversight.
What is Dual Sourcing?
Dual sourcing means contracting two vendors for the same product or service. In CDMO context, one CDMO is “primary” (majority volumes) and a second is “secondary” (reserve capacity). This model provides “insurance” against supply loss. It also fosters competition and can improve pricing.
However, it adds complexity in coordination. Executives must weigh benefits versus costs: dual sourcing is most valuable when supply interruption would be costly, e.g. late-phase or critical products.
It’s often introduced in late Phase II/III or commercial stages, after proof-of-concept is secured, because early R&D volumes are small and time-critical.
For Neuland’s API or peptide programs approaching pivotal trials, it becomes a prudent strategy. Conversely, if volume is low and the timeline is tight, a dual sourcing strategy can hurt ROI via added lead-time and management overhead.
Timing Dual Sourcing: When It Makes Sense

Industry leaders agree: dual sourcing should be timed to program maturity. Early-stage (Phase I/II) programs, with low volumes and exploratory aims, usually do not warrant dual sourcing, as it adds unnecessary work.
The sweet spot is late Phase II/Phase III and beyond, when volumes ramp, spending intensifies, and any delay threatens market launch. For fast-to-market or high-risk products, evaluate dual sourcing sooner.
Key criteria for “when to dual source” include:
- Product criticality: Essential pipeline assets or mandatory generics
- Volume growth: Increasing phase-appropriate requirements
- Lead-time sensitivity: Long assay or material lead times
- Supplier risk: Single geographic region, limited capacity, or regulatory uncertainty
How Integrated CDMOs Support Smarter Dual Sourcing
An integrated CDMO model simplifies execution within a dual sourcing CDMO strategy by reducing fragmentation across development, quality, and manufacturing functions. When one or both partners offer end-to-end capabilities—spanning development, scale-up, validation, and commercial production—coordination friction across CDMO supply chains is significantly lower.
In a well-designed dual sourcing strategy, integrated partners align documentation standards, quality systems, and process knowledge from the outset. This minimizes divergence between primary and secondary sites, reduces redundant redevelopment, and accelerates tech transfer.
For procurement and operations leaders managing dual sourcing in pharma, integration translates to fewer governance layers, clearer accountability, and stronger oversight of contract manufacturing pharma partners.
Rather than duplicating complexity, integrated CDMOs enable structured secondary sourcing within a cohesive framework—protecting resilience while maintaining operational discipline across the broader Supply chain.
Structuring a Dual-Sourcing CDMO Partnership
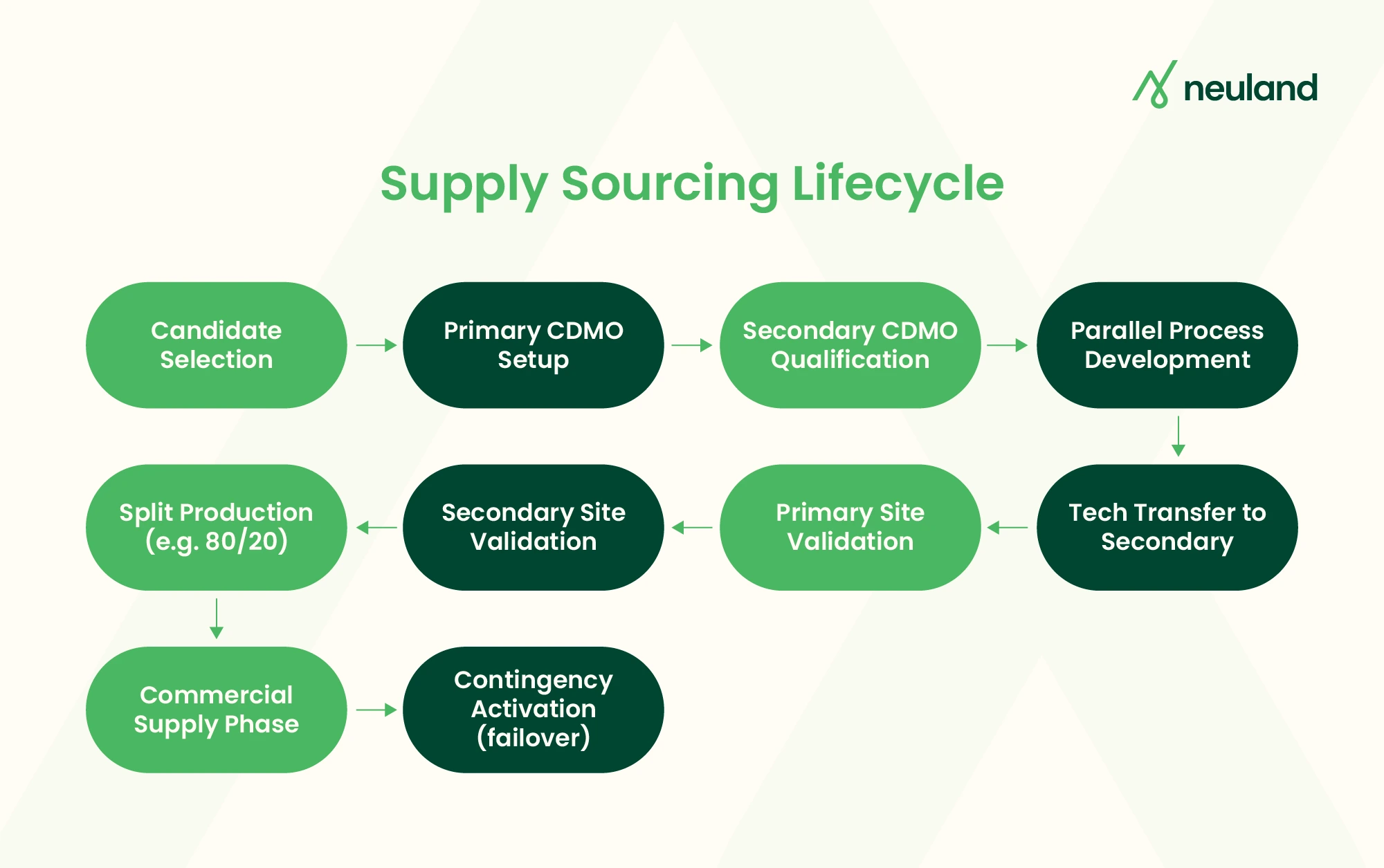
A common model is Primary–Secondary (P–S) sourcing. In P–S, the primary CDMO produces ~70–90% of supply, while the secondary maintains readiness for ~10–30%. Both suppliers meet identical quality and process specifications. This maintains production capability at the secondary without full-scale allocation. Key elements:
- Master Service Agreement (MSA): Framework contract with terms for both partners, outlining scope, IP rights, confidentiality, liability, and termination. It should allow for adding the second supplier under one overall agreement to streamline legal overhead.
- Quality Technical Agreement (QTA): FDA guidance emphasizes a QTA between sponsor and each CDMO to delineate GMP responsibilities. The QTA must clearly assign change control responsibilities to both sponsor and CDMOs. In dual sourcing, a joint QTA or mirrored QTAs ensure both CDMOs follow harmonized standards and change procedures.
- Technical Transfers: Both CDMOs must receive the same initial development data and reference materials. Common practice is to first transfer to the primary, then sequentially to the secondary. Alternatively, parallel transfers can occur if time permits, but require careful scheduling to avoid duplicated risk. Use digital PLM/QMS tools to coordinate.
- Qualification & Validation: Both sites must qualify processes (IQ/OQ/PQ). Often, the secondary qualification (Process Performance Qualification) is deferred until after the primary is validated, saving costs. However, by the time of launch both must be commercial-ready.
Governance and Coordination For Dual Sourcing in Pharma
Dual sourcing needs a centralized governance structure:
- Cross-Functional Steering Committee: Representatives from procurement, supply chain, quality, and R&D oversee the strategy. They set the dual-sourcing policy and make go/no-go decisions.
- Project Managers: Assign a dual-sourcing manager or PMO to coordinate both CDMOs. This role tracks progress, escalates issues, and ensures documentation harmonization.
- KPIs and Audits: Establish performance KPIs such as on-time delivery, process yield, deviation rates, and change request cycle time for each CDMO. Set joint review cadences (e.g., quarterly business reviews). Audit cadence should cover both sites equally. See FDA guidance for joint change control responsibilities.
- Change Control & Escalation: Any change by one CDMO (e.g., process tweak) must be communicated to the other via the sponsor. Define rules for changes (e.g., Not every minor lab change triggers cross-site change control, but major process changes do).
Dual Sourcing as a Disciplined Risk Strategy
Dual sourcing CDMO strategies should never be adopted as a reflexive policy. In stable programs with predictable demand and strong supplier performance, single sourcing may remain entirely appropriate.
However, when asset criticality, scale, or regulatory exposure increases, structured dual sourcing becomes a deliberate resilience lever within the broader supply chain strategy.
Ultimately, outcomes depend on CDMO capability and governance maturity. An experienced partner like Neuland—operating within robust quality frameworks and structured supply-chain oversight—can support resilient dual sourcing in pharma without operational fragmentation.
If you are evaluating risk exposure within your manufacturing network, now is the time to evaluate dual sourcing readiness or talk to our supply chain team about structuring a smarter sourcing model.
FAQs
|
|
|
|