CMC Development for Peptide APIs: Key Considerations
Peptide-based therapeutics are rising rapidly, offering high specificity and potency with fewer side effects than traditional small molecules.
However, developing a peptide active pharmaceutical ingredient (API) comes with unique CMC development challenges. Chemistry, Manufacturing, and Controls (CMC) in drug development encompasses all the activities that ensure a drug can be manufactured consistently at high quality and scale.
Without a well-planned CMC strategy, even a promising peptide drug can fail – not due to lack of efficacy, but because it cannot be produced reliably, safely, and in compliance with regulatory standards.
In this blog, we explore the key considerations in CMC development for synthetic peptide APIs, from process design and impurity control to regulatory requirements, and how partnering with the right contract development and manufacturing organization (CDMO) can ensure success.

Process Development and Scale-Up for Peptide APIs
Designing and scaling the synthetic route is a core element of CMC development for peptide drug substances.
Unlike small molecules produced through straightforward organic reactions, peptide manufacturing involves stepwise amino acid assembly—most commonly via solid-phase peptide synthesis (SPPS)—followed by cleavage and purification.
Even minor variations in reaction conditions at larger scale can affect final product quality, requiring careful optimization of the peptide manufacturing process, including resin selection, coupling reagents, reaction times, and effective capping strategies to limit truncation impurities.
Scaling the peptide manufacturing process adds complexity; impurities insignificant at 100 mg might become critical at larger scales if not controlled. Peptide aggregation, especially with longer or hydrophobic sequences, can reduce yield and purity during synthesis or cleavage.
Common CMC development scale-up strategies include:
- Switching to solution-phase synthesis or fragment condensation for longer peptides
- Using hybrid approaches that combine SPPS for shorter fragments with solution-phase ligation
- Conducting synthetic route scouting to identify scalable alternatives when standard routes prove impractical
Equally critical is process optimization and identification of critical process parameters. During drug substance development, peptide chemists apply design-of-experiments studies to understand variables affecting coupling efficiency, purity, and yield.
Particular attention is paid to deprotection and cleavage steps, as well as purification pooling strategies to consistently achieve target purity. Effective programs also include risk assessments to mitigate epimerization, incomplete reactions, and other peptide-specific failure modes.
Analytical and Quality Control Strategies
Analytical development is essential in peptide CMC development, as the complexity of peptide impurities and the need for exact sequence confirmation demand specialized techniques.
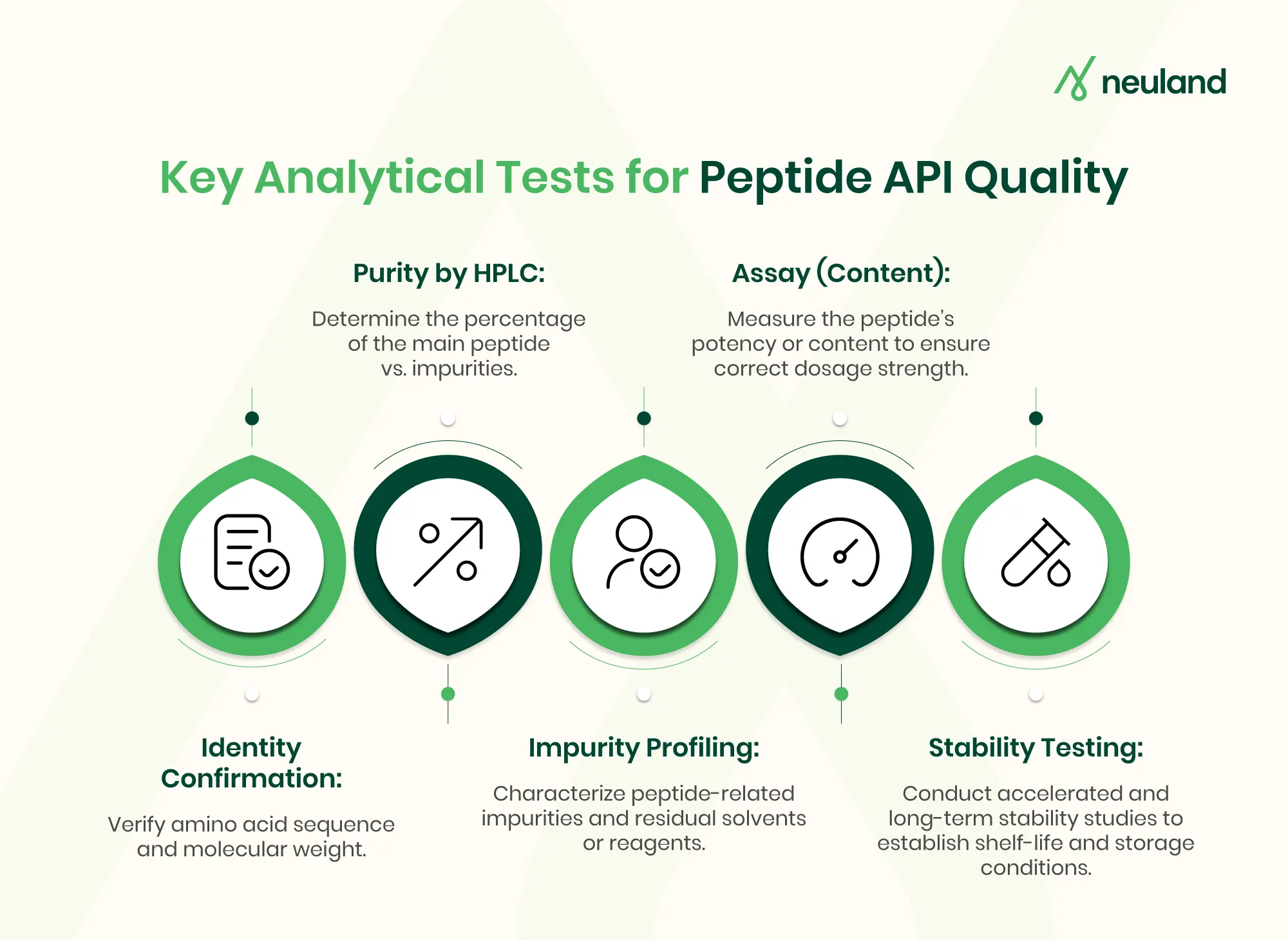
Purity is one of the most critical quality attributes in CMC development for peptides. Unlike small molecules, peptide impurity profiles are highly complex and may include truncated or deletion sequences, and degradation products such as oxidation or deamidation. These impurities can arise during synthesis, purification, or storage, particularly in solution.
Key analytical focus areas in peptide CMC development include:
- Identity and sequence confirmation using HPLC and mass spectrometry
- Detailed impurity profiling and mass balance evaluation
- Assay determination to quantify active peptide content
Beyond identity and purity, additional quality attributes must be controlled. Forced degradation and stability studies are conducted to identify degradation pathways and establish shelf-life. At Neuland, stability programs are designed to monitor purity and degradation trends over time, ensuring long-term product integrity.
All analytical activities ultimately feed into a GMP-compliant quality control program. Each batch of peptide API undergoes stringent release testing, typically including:
- Identity confirmation and purity assessment (often ≥98% for therapeutic peptides)
- Impurity limits within defined specifications
- Assay, appearance, and pH testing
A strong peptide CMC strategy relies on advanced analytical capabilities, validated methods, and stability-indicating assays that remain suitable as the process scales.
Regulatory Compliance and Phase-Appropriate CMC Strategy
Developing a peptide API must meet CMC regulatory requirements across clinical and commercial stages. Regulatory agencies require a comprehensive CMC dossier covering manufacture, characterization, controls, analytical methods, stability data, and GMP compliance as part of IND/IMPD and NDA submissions.
A phase-appropriate CMC strategy enables this alignment without unnecessary early complexity. In preclinical and Phase I development, CMC efforts focus on reproducible synthesis and essential characterization to support first-in-human studies, using risk-based approaches where methods may be qualified rather than fully validated.
As programs advance into Phase II and III, the CMC strategy expands to support scale-up, tighter controls, and regulatory readiness.
Phase-appropriate CMC progression typically includes:
- Establishing reproducible synthesis and baseline impurity understanding early
- Gradually tightening specifications and analytical validation
- Scaling manufacturing with increasing GMP rigor
- Locking and validating the process by Phase III for commercial readiness
By aligning CMC development activities with each clinical phase, clients reduce regulatory risk, avoid rework, and ensure a smooth transition from clinical development to commercialization.
Ensuring Peptide CMC Success with Neuland Labs
Selecting the right partner is critical for successful CMC development of peptide APIs. Neuland Laboratories, a global CDMO with deep expertise in peptide chemistry, supports developers across the entire pharmaceutical development process, from early process design through commercial manufacturing.
With 4 decades of experience in peptide API manufacturing under GMP, Neuland has produced peptides from milligram research scale to multi-kilogram batches using SPPS, solution-phase synthesis, and hybrid approaches. This enables scalable routes for both short and long peptides, ensuring high yield and purity as programs advance.
Quality underpins Neuland’s peptide CMC development services. Its analytical capabilities span HPLC/UPLC, mass spectrometry, impurity profiling, and reference standard qualification, supported by rigorous QC testing for identity, purity, potency, and stability.
Neuland’s regulatory CMC support ensures alignment with FDA, EMA, and ICH expectations, providing support for IND, IMPD, and NDA submissions and acting as a reliable interface with regulatory agencies.
FAQs
|
|
|
|