 Think about your last project.
Think about your last project.
How many individuals and departments participated? How many locations around the world did it involve? How smoothly did the project progress?
Could you have improved outcomes?
Pharma Projects Are Growing Increasingly Complex
In the pharmaceutical industry, projects now commonly span numerous stakeholders across multiple geographies. The challenges involved with complex project management can materialize at virtually any stage: scope management, project planning, stakeholder & team management, regulatory strategy development, or budgeting & timeline development.
Pharmaceutical API Project Management
In recent years – as constantly-changing business dynamics have combined with more complex project environments – a firm grasp of project management has become more important.
Project management in the pharmaceutical and biopharmaceutical industries is slightly unique when compared to other sectors, such as infrastructure or IT. Though the formal adoption and use of project management is a bit less mature compared to the IT sector, the challenges confronting pharma are more or less the same – and sometimes far more complex – than non-life science-related spaces.
Project management is the heart and soul of virtually every undertaking in our industry. It has become the driving force of any project to track, monitor and deliver projects within the expected timelines & budget specified. Given the risky nature of scientific research, project management is closely aligned with risk management.
Waterfall and Agile Project Management Approaches
In the lexicon of project management, there are two approaches to managing pharmaceutical research or manufacturing projects. One is the Waterfall approach, and the other is Agile management.
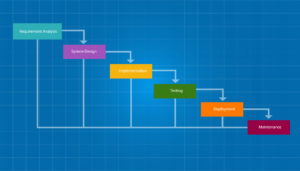
The Waterfall Development Approach
The Waterfall approach remains the pharma industry’s primary approach to product development. The name describes the methodology – phases flow downward, like a waterfall. As a methodology, it is quite appropriate to the drug industry since requirements are often static, and well-understood from the start.
In the Waterfall approach, projects consist of a series of steps or phases which must be performed sequentially. The next phase begins when the preceding phase is completed and verified. Common phases performed in order include:
- Requirements
- Design
- Implementation
- Verification
 The Agile Development Approach
The Agile Development Approach
Agile development takes a more flexible approach. Instead of delineating all of the requirements upfront (which may not be known), Agile defines each project as a set of tasks – many of which can be completed concurrently, rather than sequentially. Agile relies less on pre-planning, and more on flexibility and human interaction. Created in 2001, it was intended to streamline software development processes by de-emphasizing inefficient practices such as heavy documentation, excessive meetings, and rigid adherence to process.
In the June 2019 issue of PharmaVoice Denise Myshko discussed the rise of Agile management in the pharma space (The Agile R&D Organization), writing:
“Agile project management, first introduced in 2001, started out as a method used in software development that challenged the traditional, linear development model. The same benefits realized by the software industry, experts say, can be realized in pharmaceutical R&D.”
But – while it can help shorten drug development cycles – pharma does have some concerns with Agile. The de-emphasis on processes and tools seems to run counter to the highly-regulated nature of drug development – though it has also been pointed out that the reliance on human interactions instead “makes a robust process even stronger.”
The Right Pharma API Project Management Approach: What’s the Verdict?
Circumstances often dictate necessity. Different project management methodologies will appeal to different project types, timelines and other factors. Agile management in pharma – while lacking some of the robust documentation and other processes common to the industry – is growing in response to the need for more responsive approaches to address project uncertainties and the quest for shorter development cycles.
Neuland & the Balanced Matrix Approach
Hanover Research’s Best Practices for Matrix Organizational Structures (2013) describes balanced (or partial) matrix structures as:
“…considerably varied in form and might refer to a “temporary interdisciplinary task force for a specific purpose or a semi‐team structure developed in only part of the organization around certain functions or projects that need a high level of communication or coordination.”
At Neuland, we’ve adopted a balanced matrix design for project management, ensuring that technical leaders serve as the Project Managers (PMs). The balanced matrix gives Neuland the flexibility to create interdisciplinary, cross-functional teams, the need for which is dictated by the specifics of each project.
Although we follow the ‘Waterfall’ model (in which project planning, timelines and schedules are determined initially), we also utilize an ‘agile’ project methodology when appropriate. This allows us the flexibility to respond to issues as they arise during a project.
These two different approaches are not incongruous, and the end outcome remains the same regardless of approach: a safe and efficacious therapeutic. One method allows us to thoroughly plan out our static projects (ones with low likelihood of scope changes). The other method, well-suited to some of our earlier-stage projects, gives us (and the customer) the flexibility to adapt to changing or unpredictable circumstances, as well as tight deadlines.
Project Management Tools
To monitor and track the progress of our projects, Neuland uses the project management tool Concerto (which utilizes MS Project for the initial project planning). All project activities are sequenced or planned in parallel, depending on their dependencies. At the outset of every project, the following steps typically occur:
- Project kickoff and charter development.
- Project planning workshop, where timelines are discussed, debated, negotiated and finalized to fit the customer’s due date.
- Task & task manager determination.
Our Project Management Office
Neuland’s Project Management Office (PMO) is made up of two groups: our project proposal team and our Project Coordinators (PCs).
Among their tasks, PCs manage project scope changes. The flexibility to manage project modifications is a necessity – especially in R&D and early phase projects. Projects shift over time and the earlier the project stage, the more likely it will see changes, additions or diversions over time.
You might be thinking since scope changes can and do happen, how are they typically handled at Neuland?
Below is our process for altering projects and communicating with customers:
- Project managers and coordinators along with the business development manager are the gatekeepers of scope changes. They communicate with both the customer and the business development manager if any work requested by the customer is outside the existing project scope.
- The scope change and its impact on the initial project timelines – and also the impact on the project cost – is evaluated and communicated to the customer for approval.
- After the customer indicates acceptance of the scope change, and the project owner and the Head of R&D approve the changes, the due dates are changed in the Concerto project management system.
Do you have an upcoming project? Contact us to find out how we can help ensure your project’s success.










