I’ve mentioned the importance of route scouting in the past. Now I’d like to share some of the properties of API synthesis that can affect the yield, purity, and delivery date of a final product.
The ultimate goal is to synthesize the API product with high yields, ease of isolation from impurities, and no off-target hazardous impurities. Final products with these characteristics are more likely to pass stringent FDA and EU GMP requirements.
- Type of synthesis directly affects your yield, delivery date, and purity.
Linear synthesis of the API consistently modifies each step in a sequence. In a multi-step synthesis with yields of 80% at each step, the overall yield drops by 20% for each individual step. Thus, the total yield of a 6-step process is approximately 26.2% and yield for an 8-step process is 16.8% (Table 1).
The unreacted intermediates, B, C, D, E, F, G and H in our example below need to be purified from intermediate(s) or finished products of each reaction.
Table 1. Comparison of yields and timing of steps between linear and parallel routes of synthesis
| Linear synthesis | Parallel synthesis | ||||
| Route Steps | 80% yield for each step | Route steps | 80% yield for each step | ||
| 1 | A → B | 80.0% | 1 | A → B | 80.0% |
| 2 | B → C | 64.0% | 1 | C’ → D’ | 80.0% |
| 3 | C → D | 51.2% | 1 | E’ → F’ | 80.0% |
| 4 | D → E | 41.0% | 1 | G’ → H’ | 80.0% |
| 5 | E → F | 32.8% | 2 | B + D’→D | 64.0% |
| 6 | F → G | 26.2% | 2 | F’ + H’→J’ | 64.0% |
| 7 | G → H | 21.0% | 3 | D + J’ → J | 51.2% |
| 8 | H → J | 16.8% | |||
| Total yield | 16.8% | 51.2% | |||
Parallel synthesis reduces the number of steps that are dependent on receiving an intermediate by first preparing each part of the API molecule. The intermediate molecules are then reacted in a predesigned sequence that synthesizes the complete product.
The steps leading to the intermediate molecules dictate the structure of the impurities. The selection of the intermediate molecules and their route steps can minimize the possibility of off- target synthesis of hazardous contaminants.
The intermediates may differ in their charge properties, which allow the intermediates to be easily isolated from impurities with a single column pass. Often complex molecules have mostly positive or mostly negative or mostly neutral (hydrophobic) charges in different regions. Synthesizing each distinctly charged part separately as intermediates may simplify the isolation strategies and synthetic steps. Intermediates can react at the correct position on a core structure or intermediates can be attached to each other at the correct position until the final product is produced.
- Propinquity
As you can see in Table 1, the Propinquity or number of steps in the synthesis route directly affects the yield. Since each step has its unreacted components, the number of steps also affects the number of isolation steps and impurities. The number of steps that can be done simultaneously by different chemists can reduce delivery time, whereas downstream steps cannot be started until the intermediate(s) are done and purified.
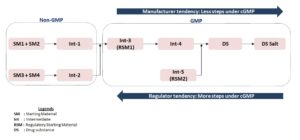
Generally, manufacturers tend to prefer developing synthetic processes under non-GMP regulations, while manufacturing APIs under current Good Manufacturing Practices (cGMP). This method – which aims to reduce the number of steps – is preferred by manufacturers for three reasons:
- Reduces costly GMP manufacture
- Reduces reporting of process variation and optimization
- Increases flexibility of process and sourcing
Regulators tend to request more steps in the GMP manufacturing process for three main reasons:
- Minimizes quality risks
- Controls impurities from starting material and subsequent steps and to ensure identity and purity of drug substance
- Purges related impurities with multiple purification steps.
At Neuland, we manufacture all of our in-house API and Custom APIs/Intermediates at our U.S. FDA approved cGMP manufacturing units.
- Complexity of structure
In general, a more complex structure usually involves more synthetic steps for the API. As discussed in our post on Benefits of Synthesis Scouting, Neuland’s Development Team can collaborate with your team – tapping extensive experience and knowledge to potentially combine steps, use parallel synthetic routes for intermediates, and reduce the delivery time while improving the final product purity.
The manufacturing of some pharmaceuticals results in isomers. Isomers are like a right wrist and a left wrist—the bending of the wrist and its ability to rotate are critical for it to function optimally … similar to complex molecules. If an intermediate step yield isomers, the active isomer may be more easily isolated in some cases.
Parallel synthesis helps reduce the number of potential isomeric variants. For example, Labetalol has four isomers in its racemic mixture. Two are relatively inactive. The potent alpha blocking activity comes from the (S,R) isomer component and the potent beta blocking activity derives from its (R,R) isomer.
- Cost Efficient Synthesis
Synthetic route planning is critical to maximizing yields. As shown in Table 1, parallel synthetic strategies overlap the scheduling of the synthesis of intermediates, and can reduce cycle time, and minimize the number of downstream processing steps.
- Carryover of impurities into drug substance
Synthetic routes should be utilized that minimize or eliminate any potential off-target synthesis of hazardous or unknown impurities. Green chemistry is important to us at Neuland, so we also plan our routes to minimize effluent generation. This can reduce the time or even the need to concentrate the intermediates.
- Minimum isolation steps in situ
Typically, synthetic routes are designed to utilize single or two isolation steps for the intermediates. By making intermediates in a parallel manner, specific columns such as specific anion or cation columns can be used more easily for their isolation. Due to efficient stepwise isolation of intermediates, the isolation of the final product is less cumbersome and generally has a very high purity.
To learn more about optimizing these 6 properties of API synthesis, contact us.